
Step Six : If the electron under considerration belongs to a d or f orbital then the shielding contribution by the electrons on the left would be 1.0 each. And all electrons further left would contribute an amount of 1.0 each. The escalation of nuclear capital costs in. Step Five: If the electron under consideration belongs to an s or p orbital then the shielding contribution by the electrons having a principal quantum number less by one would be 0.85 each. Interest charges and the construction period are important variables for determining the overall cost of capital. For 1s orbital the shielding contribution by each electron will be 0.30. Step Four: For each electron in the same group the shielding contribution by each electron will be 0.35 unless it is the 1s orbital. Step Three:Electrons on the right of the one under consideration has no shielding contribution (zero). effective nuclear charge and electronegativity - vertical periodic table trend. (1s) (2s2p) (3s3p) (3d) (4s4p) (4d) (4f). Start typing, then use the up and down arrows to select an option from the list. Step Two: Group the electrons in the following way: The effective nuclear charge for arsenic is 5. The effective nuclear charge is then calculated as follows. This means that the number of core electrons it has is (33 - 5) 28. Arsenic is in group 15 of the periodic table, so it has 5 valence electrons. Step One :Write down the electronic configuration. The atomic number of arsenic is 33, which means it has 33 protons and 33 total electrons. Follow the steps below to calculate effective nuclear charge by the Slater's rule: This calculator is based on the Slater's rule of calculating effective nuclear charge. Each change in shell number creates a new group the s and p subshells belong to the same group, while the d and f orbitals belong to their own. 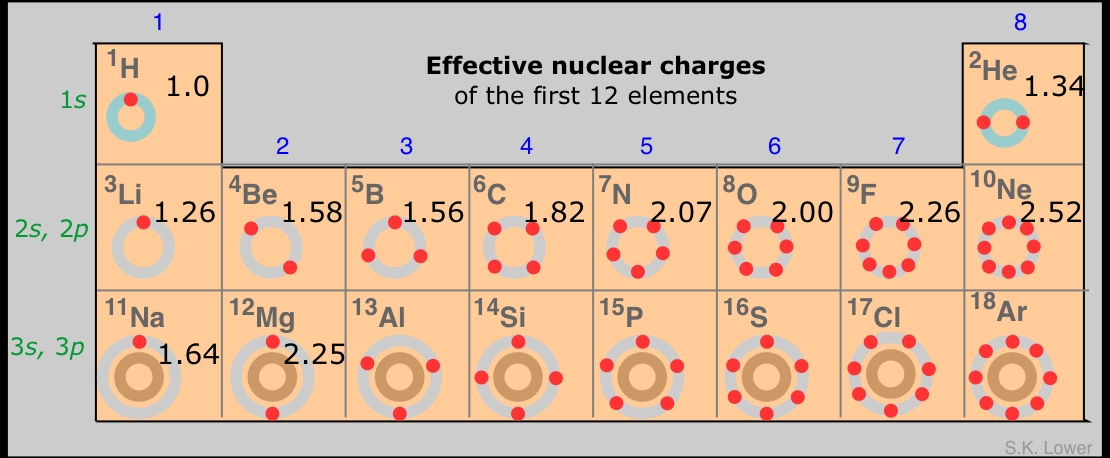
To calculate, well write out an atoms orbitals and divide them into ‘groups’. This online chemistry calculator calculates the effective nuclear charge on an electron. The effective nuclear charge is denoted by Z eff.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
